Dentistry's new frontier: the treatment of obstructive sleep apnea
By Bryan Keropian, DDSWith the advancements in design and treatment of oral sleep appliances — and in combination with the high rejection rate of the CPAP — dentistry has moved to the head of the line in providing the most widely accepted treatment for obstructive sleep apnea and snoring on the planet. Cancer, AIDS, and obstructive sleep apnea (OSA) are the most dangerous and destructive diseases on earth. OSA is reported to occur as many as 83% of women and 92% of men.1 This phenomenon occurs nightly when the tongue falls back and blocks the airway. This means that every hour of every night there are multiple periods of no O2 to every cell in the body. These periods occur five to 100+ times an hour, for 10 to 90 seconds an episode. With insufficient oxygen, cellular regeneration is unable to transpire nightly during sleep. Poor cellular regeneration results in an individual being more susceptible to numerous pathologies anywhere in the body. This nightly cellular breakdown has a destructive effect on the health of mankind. The co-morbidities for sleep apnea are numerous.2,3,4,5 OSA is the disease that dentistry now leads the world in for accepted treatment of the afflicted.The most successful treatment for OSA is CPAP. It is the No. 1 treatment prescribed by the medical community. CPAP is highly successful when it can be used. But the big problem with the CPAP are the numerous difficulties patients have using it. The CPAP is not tolerated by up to 60%,6,7 and some estimate up to 83%, of users.8 With an inability to tolerate the CPAP, these patients find themselves at great risk in regard to their health and longevity. It is a major problem for mankind when medicine’s most effective treatment tool is rejected by 60% to 83% of the users. With the high rejection rate of the CPAP, the American Academy of Sleep Medicine designated dental sleep oral appliances as the No. 1 nonsurgical alternative for the CPAP intolerant.9 Numerous sleep appliances are available to the public and distributed through dentists. These are double-arched appliances that gain success by opening the vertical dimension and advancing the mandible. Vertical opening can range from 2 mm to15 mm. The advancement can be as much as full forward prognathic extension, with forward movement up to three-quarters of an inch. The problem presented by these appliances is that they can cause pain or malocclusion.Another dental sleep appliance called the Full Breath Solution Sleep Appliance (FBS) was introduced to the dental profession at the end of 2006.10 It is a single-arched sleep appliance that fits over the maxillary or mandibular teeth. The FBS presents a very different paradigm to treatment. Rather than pulling the tongue forward by advancing the mandible, it works by utilizing a posterior bar and tail that restrains the tongue from moving upward and backward.
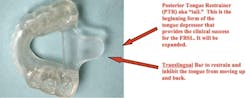
Mandibular Full Breath Solution illustrating Posterior Tongue Restrainer (PTR) and Translingual Bar The FBS has its success based on four main factors:1. The tail will be expanded to provide a tongue depressor similar to that used by a physician to view the pharyngeal airway.2. There is minimal to no advancement with the bite being set at CO or 1 mm to 2 mm anterior advancement. Treatment is successful in CO since the success of treatment is based upon restraint and depression of the tongue to open the airway, not advancement.3. The patient can open his or her mouth and have complete freedom of movement. And with all the movement, the tail still stays in the same position on the teeth, depressing the tongue.4. The FBS is small, very comfortable, and one-arched.The real success of the FBS comes with utilization of the tongue depressor and no mandibular advancement with vertical opening of 1 mm to 2 mm.Tongue depressor created with wax and then changed to acrylic on lower FBS
Wax added to tail to start forming tongue depressor.
Wax removed and acrylic added to tail. It is thinned on the superior surface so that it more comfortable and not so bulky.The Full Breath offers a number of advantages in addition to the mandibular advantages. One is no pain incited by treating in CO. The FBS exhibits clinical success above all the other appliances. In June 2009 the FBS obtained its fifth FDA government approval. In the data submitted to the FDA, there was a drop of 79% in the pre- and post-treatment AHI (number of times the airway is blocked hourly by the tongue) in patients treated with the FBS.And finally the FBS offers an avenue of remuneration for treatment unavailable with the mandibular advancers. The FBS has the posterior translingual bar and the posterior tongue restrainer/depressor that depresses and fixates the tongue. The code 41500 — Non-Surgical Fixation of the Tongue — is labeled for the procedure. In the course of treatment, we employ this procedure an average of three to six times. Reimbursement ranges from $200 to $600 every time the procedure is applied, with the average reimbursement being $400.In this time of economic pullback, we are searching for new patients, and we all know how hard it is to attract them. Ah, but there is an area that has a surplus of patients who desperately need the help of dentists: patients who suffer from snoring, mild and moderate sleep apnea, and CPAP intolerance. And the great news is that medical insurance and Medicare pay very well. Let’s review some of the statistics available to us in regard to our treatment population:
- 100 million Americans snore.
- 60% of all Americans over age 40 snore.
- 27% of all married couples snore.
- 50% to 75% of those who try the CPAP reject it.
- The CPAP industry grosses $4 billion each year.
- 20% of the grosses are for reusables, and this leaves $1.6 billion to $2.4 billion for dentistry for the treatment of apnea and CPAP intolerance.
Bryan Keropian, DDS, practices in Tarzana, Calif. He has a large, full-time sleep disorder dental practice. Dr. Keropian is the inventor of the Full Breath Solution Sleep Appliance. He can be reached at (818) 344-7200.References1. Sleep 1997; 10:705-706.2. Cardiac Consequences of Obstructive Sleep Apnea-Hypopnea Syndrome, Mt. Sinai Med. 2005; 72(1):10-12.3. Sleep Med. 2006; 7(1):73-75.4. Clin. Exp. Hypertension. 2005; 27(2-3):259-267. 5. Risk Factors and Treatment for Obstructive Sleep Apnea Amongst Obese Children and Adults, Mark Kohler, Medscape Abstract posted 2/5/2009.6. Redline S, Adams N, Straus ME, et al. Improvement of mild sleep-disoriented with CPAP compared with conservative therapy. Am Journal Respiratory Critical Care Med. 1998; 157:858-865.7. Engelman HM, Kingshott RN, Wraith PK, et al. Randomized placebo-controlled crossover trial of continuous positive airway pressure for mild sleep apnea/hypopnea syndrome. Am Journal Respiratory Critical Care Med. 1999; 159:461-467.8. Kribbs NB, Pack AI, Kline LR, et al. Objective measurement of patterns of nasal CPAP use by obstructive sleep apnea. American Journal of Respiratory and critical Care Medicine 1993.9. Kushida CA, Morgenthaler TI, Littner MR, Alessi CA, Bailey D, Coleman Jr J, Friedman L, Hirshkowitz M, Kapen S, Kramer M, Lee-Chiong T, Owens J, Pancer JP. Practice Parameters for the Treatment of Snoring and Obstructive Sleep Apnea with Oral Appliances: An Update for 2005, An American Academy of Sleep Medicine Report. Sleep 2006; 29(2). 10. Seminar, Introduction to the Full Breath Solution Sleep Appliance, At the Center for Snoring and CPAP Intolerance, Aug. 2006, Bryan Keropian DDS.