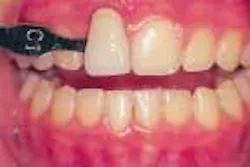
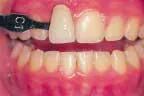
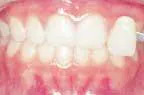
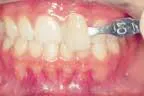
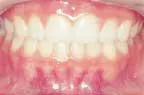
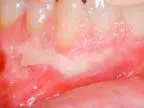
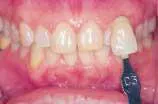
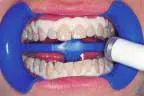
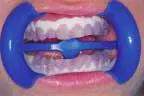
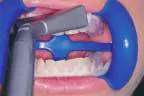
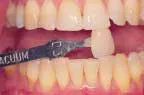
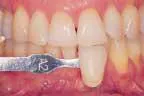
That was then ...This is now!
Tooth whitening is a commonly requested elective procedure driven by the patient's desire for a more youthful and attractive smile. Although home bleaching is considered a safe and predictable option, what about in-office bleaching and over-the-counter techniques? The public demands faster and faster results, so manufacturers have responded with a myriad of in-office bleaching systems. This article will review the current literature and attempt to clarify indications and contraindications for each technique so we can optimize our use of the latest advancements in tooth whitening.
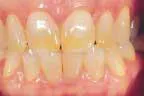
At-home bleaching has been used with great success since 19891 and provides patients with a noninvasive option to improve their smile. With the current resurgence of in-office bleaching and over-the-counter whitening systems, there is confusion regarding which approach is effective and safe. See Figures 1, 2, 3, and 4. Reviewing the literature, along with assessing advantages and disadvantages, will help define treatment parameters and manage side effects.
null
null
null
null
What are the differences between in-office and home-bleaching agents?
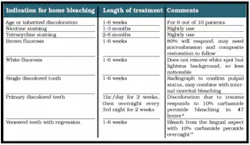
In-office whitening gels utilize 15 to 38 percent hydrogen peroxide; most systems use a 35 percent formulation. The original home-bleaching protocol proposed by Haywood and Heyman1 used 10 percent carbamide peroxide, which contains the equivalent of 3 percent hydrogen peroxide (water and oxygen) and 7 percent urea (ammonia and carbon dioxide). The active ingredient for both in-office and home bleaching is the same, hydrogen peroxide, with differing concentrations and application times. The presence of urea stabilizes hydrogen peroxide to optimize the material's shelf life and tooth-penetration characteristics. Although pure hydrogen peroxide penetrates the tooth more readily than carbamide peroxide, it is inherently unstable and dangerous to the patient when allowed to contact soft tissues. See Figure 5.
Currently only 10 percent carbamide peroxide systems have American Dental Association (ADA) approval; use of higher concentrations are without scientific basis. Higher concentrations of carbamide peroxide increase the chances of sensitivity, and their long-term stability has not been documented.
How does it work?
Bleaching teeth is not merely a surface chemical reaction, but an actual color alteration from within. Because teeth are semi-permeable membranes, hydrogen peroxide penetrates enamel and dentin to the pulp in five to 15 minutes.2 The mechanism is thought to be an oxidation reaction during which the high molecular weight and complex organic molecules cause the color of the stain to be degraded to lower molecular weight with less-complex molecules that reflect less light.3 The amount of tooth whitening achieved is related to the concentration of hydrogen peroxide and the contact time. However, there is a paucity of scientific evidence explaining what provides the inherent color of teeth and, thus, what is really changing during the bleaching process.
Safety and toxicity
There are many studies that demonstrate the safety of 10 percent carbamide peroxide with no adverse effects on the hardness or structural integrity of enamel.4,5 Laboratory assessment of the demineralization effects of 10 percent carbamide peroxide compared the effects seen with coke found that six hours of bleaching was equal to the calcium loss seen with two-and-a-half minutes of contact time with coke. Scanning electron microscopic inspection of surface enamel changes revealed some patients presented with already-roughened enamel. Future investigations should utilize prepolished samples to more accurately assess enamel surface changes after bleaching. Further investigation into possible crystalline changes in enamel and dentin when higher concentrations of carbamide peroxide are used with or without light activation is required.
Studies on the long-term safety of extended use of 10 percent carbamide peroxide in nightguard bleaching reveals no root canals, no external or internal resorption, and no permanent sensitivity.6 These patients used 10 percent carbamide peroxide in trays every night for six months to treat tetracycline staining. This seven-and-a-half-year follow-up study utilized questionnaires to document patients' tooth sensitivity and satisfaction, and compared the incidence of restorative and endodontic procedures required in the study population to the incidence seen in the general population. Tooth vitality, gum irritation, and radiographic changes were recorded six months post-treatment and on average seven-and-a-half years after active treatment.
Currently, 3 percent hydrogen peroxide (10 percent carbamide peroxide) is considered safe for lifetime use by the U.S. Food and Drug Administration (FDA), and only the 10 percent carbamide peroxide solutions are ADA-approved.7 The ADA guidelines for the acceptance of peroxide-containing products require safety data on toxicity, genotoxicity, carcinogenic potential, and two double-blind studies of two to six weeks of treatment and a two-shade color change documented for three to six months. Most studies on the carcinogenicity of hydrogen peroxide have found no evidence of harmful effects, when 0.1 to 0.4 percent hydrogen peroxide was added to the drinking water of test mice or high doses (30 percent hydrogen peroxide) were applied to animal oral mucosa. Studies citing carcinogenicity are controversial with flaws in study design where the mouse strain used already had a high incidence of carcinoma.8 In fact, the Cancer Assessment Committee of the U.S. FDA concluded that those studies could not provide adequate evidence that hydrogen peroxide was a duodenal carcinogen.9
Hydrogen peroxide is an intermediate metabolite in humans, with daily production by the liver measured at 6.48 grams. Salivary peroxidases are important to prevent adverse effects of hydrogen peroxide and they are capable of metabolizing 30 mg hydrogen peroxide in one minute. The exposure dose of hydrogen peroxide with 10 percent carbamide peroxide in dual-arch treatment has been measured at 3.52 mg, which is 0.054 percent of the daily production of hydrogen peroxide in the liver and certainly lower than the lethal toxic dose.8
How long does it take?
Every patient is different, so the length of time required to achieve the patient's desired result will vary. It has been proposed that we should match the "whites of their eyes" and "bleach until they are white."10 It is extremely difficult to stipulate bleaching time intervals and end-points as this is dependent on the patient's subjective assessment of his or her desired final color. Current assessment techniques rely on an observer using a shade guide to measure the degree of shade change. Previous follow-up studies6 have utilized patient questionnaires to record whether patients were satisfied with treatment. Most patients were happy with the color changes achieved and would recommend bleaching to other patients. The reliability of shade documentation should be improved with more objective measures, such as the use of a spectrophotometer or computer digitization in future bleaching studies. Patient compliance has been reliable with overnight bleaching and is the most cost-effective. Patient-satisfaction questionnaires would assist in future comparisons of home bleaching and in-office bleaching procedures.
null
null
null
null
null
Figure 11 Postop Vita shade D2; patient to follow with overnight bleaching with 10 percent carbamide peroxide.
About 50 percent of the product is depleted in the first one to two hours, with the other half used over the next four to 10 hours. Products designed to be used in the daytime will release the most material in the first one to two hours and may be more popular in the future due to their speed. While one in-office bleaching appointment will lighten the shade, most patients require two to six visits with an average of three in-office appointments.11 See Figures 12 and 13. Combining in-office bleaching with a home-bleaching protocol can jump-start the process for those patients willing to pay the additional fee. See Figures 6-11.
null
null
What is the status of light- and laser-activated bleaching systems?
Results from a CRA (Clinical Research Associates) study comparing Luma Arch, Opalescence Xtra Boost, and Zoom show all three systems lightened to a similar degree (1.6 to 1.7 Vitapan shade units). Investigators utilized a split-arch design in randomized order and found that the lights did not generate temperatures that increased available hydrogen peroxide, but it was the chemical additives in the in-office systems that increased degradation and reduced treatment times.15 Previous studies suggest that laser-assisted bleaching is equivalent to results seen with light-activated bleaching and 20 percent carbamide peroxide bleaching.10 Concerns regarding heat generation and its effect on the pulp require further study, as do other in-office bleaching systems. The ADA had previously approved two 35 percent hydrogen peroxide products (Union Broach and Star Brite), where one used the original floodlight to generate heat and the other product did not use a light.
Over-the-counter tooth-whitening products
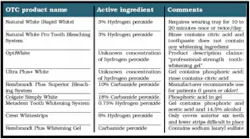
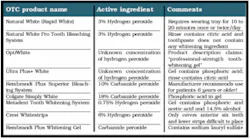
All patients requesting tooth whitening require a thorough examination and diagnosis to determine the etiology of staining and prescribe appropriate treatment. Since over-the-counter (OTC) products are classified as "cosmetics," there are less stringent guidelines and the active ingredient often is not listed. Currently, there is little data on their effectiveness. A recent article investigating the effectiveness of Colgate Simply White (18 percent carbamide peroxide) reported a 3.84 shade improvement after three weeks of use, but no pictures of the results were provided.16 Some pre-rinses and gels contain citric acid and phosphoric acid, which is of concern. One of the major drawbacks of these systems is the ill-fitting tray, which results in more ingestion of the product and prolonged treatment time due to less contact of the agent on the tooth. The popular Crest Whitestrips are only able to cover the six anterior teeth and are difficult to apply to malpositioned teeth and the lower incisors. Many whitening toothpastes do not contain a whitening agent, but merely act as stain removers.
How long does it last?
Long-term studies utilizing patient questionnaires indicate color stability for one to three years, with 74 percent of patients reporting no noticeable change after one-and-a-half years.10 After 10 years, 27 percent of patients reported no noticeable change. Because bleached teeth will eventually pick up stain and relapse, patients are told to expect touch-up treatment, where one to two nights of re-treatment is required for every week of original treatment.
Treatment protocol for sensitivity
Sensitivity may result from mechanical irritation from the tray or a chemical reaction to the whitening agent. Consider scalloping the tray material to avoid gingival contact or just scallop the facial aspect and leave the lingual area smooth for tongue contact. Trays with a reservoir avoid pinching pressure on the teeth and aid seating. Although less material may be lost and ingested when there is a reservoir, bleaching results are the same.17 Reducing the length of contact time or the frequency of application also may alleviate sensitivity problems. Active treatment of sensitivity with fluoride or potassium nitrate applied via the bleaching tray is also effective. Application of potassium nitrate for 10 to 30 minutes prior to or after bleaching, or alternating nights with bleaching material, has been demonstrated to be effective in more than 90 percent of patients.18 Fluoride acts as a tubular blocker and thus reduces fluid transmission and dentinal sensitivity. The mechanism of action of potassium nitrate is similar to an anesthetic wherein the transmission of nerve impulses is affected.
Conclusion
While dentist-supervised home bleaching is a predictable option for many patients desiring tooth whitening (see Figures 14 and 15), newer, faster systems warrant further investigation. Further long-term case controlled studies are required to evaluate the use of higher concentrations of carbamide peroxide and its effect on tooth structure and restorative materials. The seven-and-a-half-year follow-up study on patients who used 10 percent carbamide peroxide for six months to treat tetracycline staining indicated no increased incidence of restorative and endodontic treatment.6 Although evaluation of patient satisfaction has been documented, ongoing comparison of various bleaching techniques is required. Until we understand all of the factors contributing to tooth color and discoloration, it remains difficult to accurately recommend definitive time intervals for initial bleaching and future touch-up treatment.
null
null
In-office bleaching holds promise and is a viable option for patients who prefer to shorten the treatment time. Over-the-counter options need to be approached with caution in cases where patients using these products are undiagnosed and unsupervised. Tooth-whitening materials and techniques are evolving at a rapid rate. Ongoing research into their long-term safety, longevity, and patient satisfaction is required so clinicians can have a scientific basis for recommending and supervising this conservative treatment option for patients.
Jean C. Wu, DDS
Dr. Wu maintained a private prosthodontic practice in Australia, and now practices in Newport Beach, Calif. She is on faculty with the Newport Coast Oral Facial Institute and is involved with research projects on dental implants and materials. Contact Dr. Wu at (949) 760-6288 or [email protected].
Cherilyn G. Sheets, DDS
Dr. Sheets maintains a private practice in Newport Beach, Calif., with an emphasis on aesthetic and restorative dentistry and implants. She is co-executive director of the Newport Coast Oral Facial Institute. You may contact her at (949) 760-6288 or [email protected].
Jacinthe M. Paquette, DDS
Dr. Paquette is a prosthodontist specializing in aesthetic, implant, and reconstructive dentistry and occlusion and Temporomandibular disorders. She is co-executive director of the Newport Coast Oral Facial Institute. Contact her at (949) 760-6288 or [email protected].
References —
- Haywood VB, Heyman HO. Nightguard vital bleaching. Quintessence Int 1989;20:173-176.
- Cooper S, Bokmeyer TJ, Bowles WH. Penetration of the pulp chamber by carbamide peroxide bleaching agents. J Endod 1992;18(7):315-317.
- Nathoo SA. The chemistry and mechanisms of extrinsic and intrinsic discoloration. JADA 1997;128:6S.
- McCracken MS, Haywood VB. Demineralization effects of 10% carbamide peroxide. J. Dent. 1996;24:395-398.
- Haywood VB, Leech T, Hayman HO, Crumpler D, Bruggers K. Nightguard vital bleaching: Effects on enamel surface texture and diffusion. Quintessence Int 1990;21:801-806.
- Leonard RH, Haywood VB, Caplan DJ, Tart ND. Nightguard vital bleaching of tetracycline stained teeth 7.5 years post-treatment. J Dent Res 2002;81(#1962).
- ADA Guidelines for the acceptance of peroxide containing oral hygiene products. JADA 1994;125.
- Yiming L. Peroxide-containing tooth whiteners: An update on safety. Compendium 2000;21(28):S4-S8.
- Food and Drug Administration. Hydrogen peroxide: proposed affirmation of GRAS status as a direct human food ingredient with specific limitations. Federal register 1983;48:52323-53333.
- Haywood VB. Supervised at-home bleaching is safest, most effective-An interview. Dental Products 2000;82-91.
- Haywood VB. A comparison of at-home and in-office bleaching. Dentistry Today 2000; 19(4):44-53.
- Brantley DH, Barnes KP, Haywood VB. Bleaching primary teeth with 10% carbamide peroxide. Pediatric Dentistry 2001;23(6)514-516.
- Haywood VB, Parker MH. Nightguard vital bleaching beneath existing porcelain veneers: A case report. Quintessence In 1993;3(11):743-747.
- Haywood VB. Greening of the tooth-amalgam interface during extended 10% carbamide peroxide bleaching of tetracycline stained teeth: A case report. J Esthet Restor Dent 2002;14;12-17.
- New generation in-office vital tooth bleaching, Part 1. CRA Newsletter 2002;26(11):1-3.
- Nathoo S, Stewart B, Zhang YP, Chaknis P, Rustogi KN, DeVizio W, Petrone M, Volpe AR. Efficacy of a novel, nontray, paint-on 18% carbamide peroxide whitening gel. Compendium 2002;23(11):26-31.
- Matis BA, Yousef M, Cochran MA, Eckert GJ. Degradation of bleaching gels in vivo as a function of tray design and carbamide peroxide concentration. Operative Dentistry 2002;27:12-18.
- Haywood VB, Caughman WF, Frazier KB, Myers ML. Tray delivery of potassium nitrate-fluoride to reduce bleaching sensitivity, Quintessence Int 2001;32:105-109.