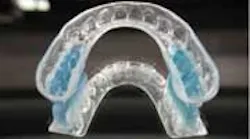
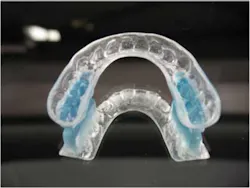
First-time clinical study tests a noninvasive treatment for Tourette's using an oral orthotic device
Georges Gilles de la Tourette, a French physician and neurologist, first diagnosed Tourette’s syndrome in 1885. Research in 1972 argued that Tourette’s is a neurological disorder, not a psychological disorder. Since the 1990s, Tourette’s has been considered a neuropsychiatric condition.
Tourette’s usually has a childhood onset, characterized by multiple motor tics and one or more vocal tics. Tics are sudden, repetitive vocalizations and nonrhythmic movements of the body. They typically wax and wane, and are usually preceded by a premonitory urge.
ADDITIONAL READING |Clinical study researches use of oral appliances in mitigating severity, frequency of motor and vocal tics
With poor or inadequate management of these symptoms, patients are often socially isolated, emotionally distressed, have difficulty in school, and problems keeping a job or sustaining a career. Some experience physical pain from intense and complex tics.
Current treatments consist of antipsychotic and adrenergic agonist medications, Comprehensive Behavioral Intervention for Tics (CBIT) therapy, and Deep Brain Stimulation with varied responses patient by patient.
This syndrome currently has no cure, and the etiology is still unclear in the medical community. However, it is the intention of our study to provide medical and dental teams with the ability to mitigate the clinical manifestations of Tourette’s.
This is the first time a clinical study of this type has been done testing a noninvasive method of treatment for Tourette’s. Funded with $340,000 from TicTocStop, the study began in September 2014 and will run through mid-2015.