News you can use
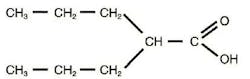
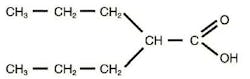
Use of Anti-Epileptic Drug during Pregnancy Associated with Increased Risk of Autism Maternal use of valproate, a drug used for the treatment of epilepsy and other neuropsychological disorders, during pregnancy was associated with a significantly increased risk of autism in offspring, according to a study in the April 24 issue of JAMA.(1,2) Antiepileptic drug exposure during pregnancy has been associated with an increased risk for congenital malformations and delayed cognitive development in the offspring, but not much is known about the risk of other serious neuropsychiatric disorders.(3) Genes play a significant role in the risk of autism, and disease-related genes may be identified in up to 25% of children with autism.(4) However, a variety of environmental factors may increase the risk of autism, and prenatal valproate exposure would be a modifiable environmental exposure.(5)
The main outcomes and measures of the study were absolute risk (cumulative incidence) and the hazard ratio (HR) of autism spectrum disorder and childhood autism in children after exposure to valproate in pregnancy. The conclusion was that maternal use of valproate during pregnancy was associated with a significantly increased risk of autism spectrum disorder and childhood autism in the offspring, even after adjusting for maternal epilepsy.(1) For women of childbearing potential who use antiepileptic medications, these findings must be balanced against the treatment benefits for women who require valproate for epilepsy control.Higher Blood Levels of Omega-3s Linked to Lower Mortality Risk High plasma levels of long-chain omega-3 fatty acids are associated with reduced mortality, especially cardiovascular mortality.(6) A total of 2,700 older U.S. adults without heart disease underwent serum measurements of omega-3 fatty acids and then were followed for about 15 years. During that time, there were 1,625 deaths. In adjusted analyses, participants with the highest baseline levels of omega-3s had a 27% reduction in total mortality risk, relative to those with the lowest levels. A decrease in cardiovascular mortality, particularly arrhythmic cardiac mortality, was responsible for most of the risk reduction.
The researchers note that "the observed mortality differences corresponded to approximately 2.2 more years of remaining life after age 65 years" in those with the highest omega-3 levels. They say their findings suggest that dietary omega-3s in older age might reduce overall mortality.(6) Significant controversy exists regarding the association of omega-3 polyunsaturated fatty acids (PUFAs) and major cardiovascular end points.
A study in 2012 assessed the role of omega-3 supplementation on major cardiovascular outcomes.(7) It was a systematic review and meta-analysis. The conclusion was that omega-3 polyunsaturated fatty acids supplementation was not associated with a lower risk of all-cause mortality, cardiac death, sudden death, myocardial infarction, or stroke based on relative and absolute measures of association. The authors concluded that the findings do not justify the use of omega-3 as a structured intervention in everyday clinical practice or guidelines supporting dietary omega-3 administration. We now have a new study that sheds more light on this topic.(6)American Academy of Neurology Issues Guideline on Treatment of Neurocysticercosis And last, but not least, The American Academy of Neurology (AAN) has released an evidence-based guideline on treating neurocysticercosis, a tapeworm infection causing seizures that is common in developing countries and is now on the rise in developed countries, including the United States. The guideline is published in the April 9, 2013, print issue of Neurology, the medical journal of the American Academy of Neurology.(8) This previously rare disease in the United States is increasingly prevalent, and the infection and epilepsy due to the infection are preventable.
The guideline states that a combination of the drug albendazole and a corticosteroid can effectively treat neurocysticercosis. Albendazole is used to kill the parasite, and dexamethasone or prednisolone is used to treat the inflammation that develops as the parasite is dying. The tapeworm Taenia solium can infect people in two ways: the infection of the intestines happens when a person eats raw or undercooked meat that has the tapeworm cysts in it; a person gets neurocysticercosis by eating food or drinking water contaminated with the tapeworm eggs. Food can be contaminated with fecal matter from a person who has the infection. Usually, this happens from poor hygiene within a household or other environment. Infection from this tapeworm is preventable. Good personal hygiene is critical to prevent the transmission of infection from person to person. This involves washing hands thoroughly after using the bathroom.References 1. Christensen J, Grønborg T, Sørensen M, et al. Prenatal Valproate Exposure and Risk of Autism Spectrum Disorders and Childhood Autism. JAMA. 2013; 309(16):1696-1703. doi:10.1001/jama.2013.2270. 2. http://www.drugs.com/cdi/valproate.html. 3. Meador KJ, Baker GA, Browning N, et al; NEAD Study Group. Fetal antiepileptic drug exposure and cognitive outcomes at age 6 years (NEAD study): a prospective observational study. Lancet Neurol. 2013;12(3):244-252. 4. Miles JH. Autism spectrum disorders—a genetics review. Genet Med. 2011;13(4):278-294. 5. Gardener H, Spiegelman D, Buka SL. Perinatal and neonatal risk factors for autism: a comprehensive meta-analysis. Pediatrics. 2011;128(2):344-355. 6. Mozaffarian D, Lemaitre RN, King IB, Song X, Huang H, Sacks FM, Rimm EB, Wang M, and Siscovick DS. Plasma Phospholipid Long-Chain ω-3 Fatty Acids and Total and Cause-Specific Mortality in Older Adults: A Cohort Study. Ann Intern Med. 2013;158 (7):515-525. 7. Rizos EC, Ntzani EE, Bika E, Kostapanos MS, Elisaf MS. Association Between Omega-3 Fatty Acid Supplementation and Risk of Major Cardiovascular Disease Events: A Systematic Review and Meta-analysis. JAMA. 2012;308(10):1024-1033. 8. Baird RA, Wiebe S, Zunt JR, Halperin JJ, Gronseth G, Roos KL. Evidence-based guideline: Treatment of parenchymal neurocysticercosis: Report of the Guideline Development Subcommittee of the American Academy of Neurology. Neurology. 2013 Apr 9;80(15):1424-1429.