A pouch is a pouch, right?
Wrong! Here's what you need to know about sterilization pouches
By Andrew G. Whitehead
Why is a pouch not just a pouch? When it comes to sterilization packaging, in particular self-sealing sterilization pouches, many purchasing decisions are based either on price, the choices that your source offers, or a combination of both. Why? Nearly everyone believes that sterilization pouches are basically the same; in reality, they are not. In fact, many of the pouches sold in the dental industry today are not acceptable for sale in the medical industry because they do not meet medical industry safety standards.
null
What do you really know about the sterilization pouches you use? What should you know? What features should be standard in a quality pouch and why?
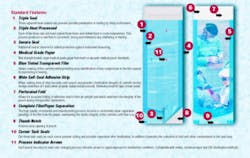
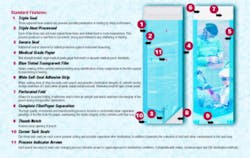
Sterilization pouches should be constructed of medical-grade surgical Kraft paper (basis weight range 37-42 pounds) and a transparent 2-mil polypropylene/ polyester laminate film. A tinted film is preferred because the tint allows any compromises in the film's integrity to be more readily visible.
The preferred pouch design should be of multiple side and end seals with a self-sealing adhesive strip that must adhere to both paper and film during closure; i.e., 50 percent paper, 50 percent film, and a perforated fold.
The combination of an adequate width self-sealing strip and a perforated fold allow for a clean, uniform fold and seal, thereby creating an effective seal. This is critical to maintaining sterility of the contents until ready for use.
If you are unsure about the effectiveness of your current pouch, simply water-test it. Seal the pouch (with nothing inside) using the self-adhesive strip. Open the chevron top seal of the pouch just enough to fill halfway with water. Observe the pouch ... Does it leak water? If it does, you are not getting a proper seal with your current pouch. This is caused either by poor user technique or poor quality pouch design in the seal area (e.g., adhesive is sealing to the film instead of paper and film).
Tack seals should be standard at the sealing end of all pouches. This prevents curling of the film after sterilization. No curls mean there is no place for dust to collect while the pouch is stored. Remember, the pouch is opened from this end, and any dust particles that may collect in this area will fall onto the pouch contents when opened.
A thumb notch also is important. While not mandatory, it is preferable, because it greatly enhances the hand gripping of the paper and film for proper opening of the pouch. The correct protocol for removing the contents from a pouch is to separate the film from the paper, not to push the contents through the film or paper, thus rendering the contents nonsterile.
It is extremely important to the medical profession that the film separates from the paper when the pouch is opened. If any portion of the film remains attached to the paper when opening (separating) the film from the paper, the contents are considered to be contaminated and must be resterilized. Why? The film that remains attached to the paper has been exposed to day-to-day elements in the air for as long as the pouch has been stored since sterilization. Should any of the contents come in contact with the film upon being picked up, they are considered to be contaminated.
While the complete separation of film from paper is a must in the medical industry, it is relatively unknown to the dental industry. This is why many pouches sold to dentists cannot be sold to the medical industry. To test the separation quality of your pouch, simply separate the film from the paper at the tack seals, grasping the film in one hand and the paper in the other, and completely separate. If any film remains on the paper, repeat the process with another pouch. If you continue to leave any film on the paper, the quality of the pouch is questionable.
How does your current pouch measure up? What features does your current pouch have or, more importantly, what features does it lack? Does the film completely separate from the paper? Perhaps a pouch is not a pouch after all. Become aware of the differences that exist and give careful consideration when making your next purchase.
Another important factor is size. There is no industry standard for identifying the size of a pouch. In many instances, one company's 3 1/2" x 9" pouch is as large or larger than another company's 3 1/2" x 10" pouch. The amount of usable space is what counts. Measure the usable width from inside seal to inside seal and the length from the end of the film to the inside seal at the unopened end of the pouch. These are the measurements that really matter. Choose the size the best matches the size and shape of the contents. Overloading or using an undersized pouch can make it impossible to achieve a proper seal. Any misalignment at the seal interface can create gaps in this critical area.
Instrument sterilization is one of the most critical processes in the dental office. Is price important? Absolutely, but so is the quality, consistency, and specifications of the pouch you use. You are not only protecting your patients, you are protecting your staff as well as your practice.
You are all worth the minimal cost difference that using a quality sterilization pouch will add to your safety. Remember, if you don't pay attention to this, no one else will. At the end of the day, the most important factor is you.
Andrew G. Whitehead is a vice president for Crosstex International, manufacturer of infection-control and disposable products distributed in 60 countries. He has more than 30 years of experience in the dental industry. He is a founding member and board member of the Organization for Safety and Asepsis Procedures (OSAP), dentistry's resource for infection control and safety. He may be reached at (631) 582-6777 or by email at [email protected].