Laser integration for optimum cosmetic dentistry
WRITTEN BY Janet Hatcher Rice, DDS
Dental lasers are becoming an integral part of contemporary cosmetic dentistry, yet they are only one tool in the dental armamentarium for cosmetic procedures. They can artistically sculpt soft tissues while providing precision, increased visibility, improved impression technique, predictable healing with decreased scarring, and reduced anesthesia and postoperative swelling1 With the addition of the hard-tissue laser, cosmetic bonding procedures are more predictable by providing enhanced bond strength, more comfortable by requiring little to no anesthesia, and less invasive via controlled removal of tooth structure.2 Dental laser technology has accumulated a wealth of scientific and clinical studies from decades of laser research.
Background
Laser research began in 1960 with the pulsed ruby laser built by Maiman3 for Hughes Research Laboratories. One year later, Goldman,4 a physician at the University of Cincinnati, began documenting the ability of various lasers to incise, coagulate, ablate, and vaporize biologic tissues, including dental tissues. In 1963, Stern and Sognnaes,5 along with Goldman, began in vitro investigations on teeth with an emphasis on their ability to melt and recrystalize enamel, which reduced enamel’s permeability to acidic oral conditions. However, by the end of the 1960s, the conclusion was that the deep penetration of the 694 nm wavelength of ruby lasers increased the chance of pulpal injury. The ruby laser was abandoned in favor of newer lasers, such as carbon dioxide (CO)2 lasers.6 These were the first lasers marketed for intraoral use; however, throughout the 1970s and the first half of the 1980s, clinical use of the CO2 laser in the oral cavity was primarily confined to oral surgeons. Acceptance was slow due to problems getting the laser light from the device to all areas of the oral cavity. In the late 1980s, medical lasers with unique delivery systems paved the way for improved access. Smaller devices and flexible delivery systems, such as optical fibers and hollow wave-guides, finally provided accessibility to the oral cavity. Research surfaced in 1988 about the ability of an erbium (Er):YAG laser to ablate dental hard tissues.
Lasers gained entry into general practice in May 1990, when the FDA cleared a pulsed Nd:YAG laser developed by Myers and Myers7 for intraoral soft-tissue surgery. The dLase 300 (Sunrise Technologies, Sunnyvale, Calif.) was the first laser for general dentistry.8 The FDA continued to give dental laser marketing clearance for various soft-tissue uses, but the breakthrough that increased the lasers’ desirability for use in general practice was in 1997, when the FDA granted approval for caries removal and cavity preparation by an Er:YAG (2.94µ) laser (Centauri Er: YAG laser, Premier Laser Systems, Irvine, Calif.). Another landmark clearance came in 2002 when the FDA approved cutting of oral osseous tissues and root canal treatment by an Er,Cr: YSGG(2.79µ) laser (Millennium Er:YSGG laser, Biolase Technology, San Clemente, Calif.). These marketing clearances, based on extensive scientific and clinical research, were granted to a device demonstrating safe and effective use for specific indications.
Education
When contemplating laser treatment, keep in mind that all lasers require specialized training and attention to safety.9 The Curriculum Guidelines for Dental Laser Education were written in 1992, have been revised,10 and were formally adopted and implemented by the Academy of Laser Dentistry (ALD). They define the standard of education for practitioners, with a goal to enhance the understanding and knowledge of laser applications in dentistry.11 The ALD believes that all those who use dental lasers should be properly trained and recommends a Standard Proficiency level (formally known as Category II).12 OSHA is concerned with worker safety and reviews laser installations, laser safety officer documents, and staff training.13
Laser selection
When integrating laser technology for cosmetics, selection of the appropriate wavelength, power, delivery system, mode of emission, handpieces, and tips is critical. Current availability of multiple devices and wavelengths makes laser selection difficult. First and foremost, selection should depend on the unique absorption characteristics of the device’s wavelength. Strong absorption by a biologic component implies that the selected wavelengths exhibit shallow penetration in that target tissue. The laser practitioner should “match” the laser wavelength to the tissue being treated.5 Sufficient power, generally expressed in watts or millijoules/second, is important. The delivery system (optical fibers or hollow wave-guides) should allow for easy access to all areas of the oral cavity. A laser device can emit light energy in one of four modes: continuous wave - the power runs continually as long as the device is activated; gated-pulsed - periodic alterations in the power by being on or off; free-running pulsed - large peak powers for extremely short periods of time, usually in preset microseconds; and super-pulsed - emits high peak powers but is changeable. The emission mode depends on the type of tissue, amount of tissue to be removed, and the practitioners’ preference and ability.
Lasers come with varying handpieces and tips that need to be evaluated and selected for appropriate clinical usage.
Clinical considerations
Dental structures - such as teeth, attached gingiva, epithelial attachment, periodontal ligament, and alveolar crest - are each important and should be evaluated carefully for composition.1 Tooth structures should be evaluated for depth of enamel and dentin and proximity of the pulp. The zone of attached gingiva must be wide enough, the epithelial attachment and periodontal ligament should be evaluated for signs of disease or trauma, and the alveolar crest should be normal and without infrabony defects. Lasers should not be used to ablate (remove) amalgam. Lasers cannot ablate gold or porcelain; however, they can ablate caries, enamel, dentin, pulp, cementum, bone, cement, composite resins, and glass ionomers. Different irradiation parameters control laser-tissue interaction. They are wavelength, continuous or pulsed emission, repetition rate, pulse duration, pulse energy, beam size and delivery method, spatial and temporal characteristics of the laser beam, and optical properties of the tissue.14
Laser use in cosmetic dentistry
Soft-tissue cosmetic laser dentistry includes the treatment of gingiva. Gingival tissues frequently need to be altered because of inflammation, proliferation, subgingival restorations, subgingival caries, or irregular contours. The finish line for crowns, veneers, or bonded restorations may need to be placed into the gingival sulcus, which can cause bleeding. A hemorrhage in the gingival sulcus can make impression making or bonding procedures impossible and healing unpredictable. In these cases, laser sulcular gingivoplasty can be used to develop a new gingival sulcus, control hemorrhaging, and provide esthetic gingival contours.1
Creating a healthy new zone of attachment will enhance the long-term prognosis for cosmetic dentistry. Hard-tissue cosmetic laser dentistry is useful when tooth structure needs to be restored. Cases of cervical erosion caused by abrasion or abfraction require minimal preparation but need maximum retention. In these cases, laser tooth ablation and surface modification will conservatively remove and “etch” the affected tooth structure in an irregular fashion, which is ideal for the placement of composite or glass ionomer restorations and cements.15 Sheer bond strength is greater in the laser-prepared cavity combined with acid etch than etching alone.16
Laser sulcular gingivoplasty
When considering a smile’s components, dentists focus on color, shape, alignment, and function. The quality of the gingival architecture is extremely important but often overlooked. Healthy, symmetrical gingiva plays an important role in the creation of a beautiful smile.17 A dental laser that has a good absorption characteristic for hemoglobin is the best for developing a healthy sulcus. This author prefers diode lasers, which use fiber-optic delivery and direct contact.
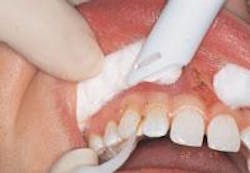
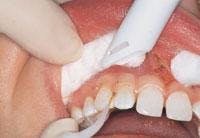
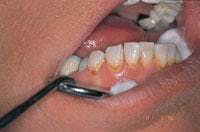
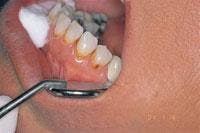
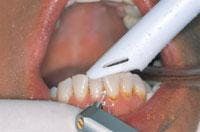
Laser fibers 400 to 600 µ in diameter are ideal in spot size for this procedure. The quartz glass fiber must be cut properly to permit complete transmittance of laser light through the fiber. High-volume evacuation should be used to remove the plume (smoke and biologic debris) and cool the treated area. The handpiece should be lightweight, comfortable to grasp, sterilizable, and should accept disposable, angled plastic tips. Angled tips are necessary to contour gingiva in an esthetic, rounded shape. A feather-light stroke with quick, deft strokes should be used. The laser handpiece should keep moving to prevent unwanted thermal damage to underlying tissues. The operator must “feel” the tissue being removed and adjust the speed of the stroke to the removal rate. The operator should not attempt to remove the tissue by dragging the glass fiber through it, because this causes bleeding. The fiber should be held parallel to the long axis of the tooth as much as possible to prevent exposure of the root surface to the laser energy (Figure 1). Hemostasis occurs because of the capacity of the laser to seal small blood vessels as a result of the photothermal interaction with the hemoglobin.
The current gingival sulcus should be measured with a periodontal probe. Unless a large amount of tissue needs to be removed, it is best to start at the gingival margin and work apically until the desired amount is removed and esthetic contour is achieved. A different technique should be used when developing a sulcus for crown and bridge or implants. After preparing the tooth or implant abutment, the operator should start at the bottom of the sulcus and seal the most apical areas. Only if removal of excess tissue is required should the sides be sealed. The operator should stop periodically and remove debris from the fiber tip with water-moistened gauze. If carbon (char) builds up on the tissue, this should also be wiped off to keep heat from building up.
Postoperative instructions should include warm saltwater rinses and use of an ultra-soft toothbrush. Over-the-counter pain medications, such as nonsteroidal anti-inflammatory drugs, may be beneficial.
Laser tooth surface
ablation and modification
Dental enamel is the hardest tissue in the body, and controlled cutting of enamel with lasers was an elusive goal for many years. Current laser technology allows for precise removal of small amounts of enamel and dentin using a photoacoustic action called ablation. All dental hard tissue contains various amounts of water. When laser light hits hard tissue, the water is heated in a small area. Acoustic shock waves are produced due to the rapid volumetric expansion that occurs when water changes from liquid to gas. This expansion causes the surrounding tooth structure to explode. The water spray of the handpiece accelerates this effect by removing exploded tissue while transferring minimal heat to the remaining tooth.15
Erbium lasers have strong absorption in water and hydroxyapatite and are most efficient for ablation and modification. The pulsing action creates a surface effect that resembles chemical etching, with an expanded surface area and increased surface energy. When acid and laser modification are combined, the bond strengths with restorative resins are greater than with either method alone.16
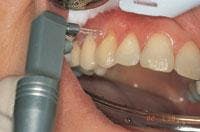
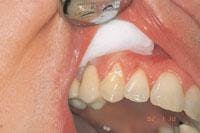
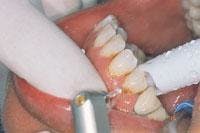
Starting with the manufacturer’s recommended power setting (millijoules) and pulses per second (pps), treatment should be initiated perpendicular to and 1 mm above the surface of the target (Figure 2) unless a contact tip is used. Anesthesia is rarely needed for this technique, which is amazing, since profound anesthesia is needed for all Class V preparations with a high-speed handpiece. The laser makes distinct, circumferential marks and “pop” noises as it removes tooth structure (Figure 3). The operator should gauge the removal rate by how quickly the marks are made and how loud the pops are, and then inspect what has been removed. Even with contact tips, the “feel” of tooth removal is different. The laser removes tooth slower than a high-speed handpiece,19 so the laser handpiece must be moved more slowly and deliberately than a drill. Laser energy is emitted from the tip only and should be directed perpendicularly to the target. When preparing occlusal surfaces, the operator should not start in the pit and fissures as in conventional drilling because it is difficult to hold the tip perpendicular. He/she should start away from the pit at the cavomargin where the prep will end, direct the water stream to the target tissue, and keep the operative site wet. This is another reason not to begin at the pits and fissures, because water cannot enter the pits well and the heat buildup will cause pain.
Cavity preparation should be based on physical and mechanical principles that will enhance the chemical bond. G.V. Black preparations are no longer needed with today’s bonding technology. In fact, the rounded shapes and punctive surfaces are perfect for adhesive dentistry. The physical properties and capabilities of the different restorative materials and their responses to stress must be appreciated, particularly in Class V restorations. With this in mind, think of all the cases of abrasion, abfraction, and incisal wear that could benefit from this technology, especially in the aging baby boomer population! These areas can be glassy smooth, highly polished, and difficult to restore. These patients have already lost valuable tooth structure, and high-speed drilling only adds insult to injury. Laser tooth ablation and surface modification provides a less invasive, more comfortable, cosmetic restorative alternative to conventional modalities.
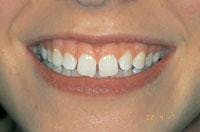
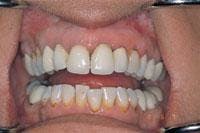
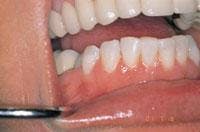
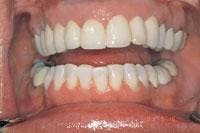
An 18-year-old female presented for an initial comprehensive oral exam. She was concerned about the appearance of her teeth and gums and wasn’t happy with their color, shape, and gingival contour. The preoperative clinical photograph (Figure 4) revealed multiple spaces, malalignment and uneven gingival heights. Treatment included full facial composite bonding on Teeth Nos. 7 through 10 and laser sulcular gingivoplasty on Teeth Nos. 6, 7, 10, and 11. A laser frenectomy was also performed. Other options were discussed but due to the patient’s age and lack of interest in surgery or orthodontics, this was considered optimum treatment. Local anesthesia, one carpule of Septocaine 1:100,000 epi, was infiltrated bilaterally in the buccal fold.
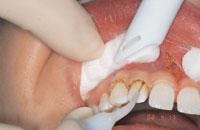
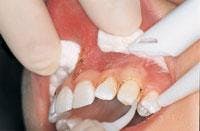
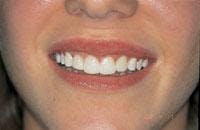
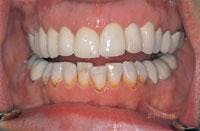
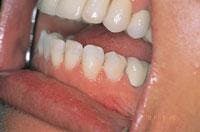
Treatment was initiated with periodontal probing from Teeth Nos. 6 through 11. Only 2 mm was available for removal on Teeth Nos. 6 and 11 and 3 mm on Tooth No. 7. Tooth No. 10 was only slightly treated to round out the cleft at the top of the gingiva and the tapered sides. The Opus 10 (Lumenis, Santa Clara, Calif.) 830nm-diode laser with a 360-micron fiber was used at 1.4 watts continuous wave. During this procedure, photos were taken demonstrating the correct technique of holding the fiber parallel to the long axis and working from the gingival margin apically (Figures 5 and 6). Composite bonded restorations were adapted to the facial surfaces of Teeth Nos. 7 through 10. An immediate postoperative photo was taken (Figure 7).
Case report No. 2
A 39-year-old female patient presented for an initial comprehensive oral exam. She wasn’t satisfied with her current crowns and was unhappy with teeth color, shape, and gum recession (Figure 8). The patient was treatment-planned for porcelain-fused-to-high-noble-metal crowns from the maxillary right first bicuspid to maxillary left cuspid with laser sulcular gingivoplasty with a diode laser. She was also displeased with the lower teeth and thought she would need to have them crowned. Instead, the lowers were treatment-planned for buccal composites using laser tooth ablation and surface modification and incisal edge reshapement, all of which could be done at the cementation appointment with no anesthesia. The patient was ecstatic and could hardly believe everything could be done in two appointments. After preparing the maxillary teeth, the patient returned six weeks later for the final cementation (Figures 9, 10, and 11). There was superior healing of tissue around the maxillary crowns. The lower bicuspids had significant decay but no anesthesia was used. The mandibular teeth were prepared with an erbium laser, the Opus 20 (Lumenis, Santa Clara, Calif.) Er:YAG laser at eight pulses per second and 800 millijoules or 6.4 watts (Figures 12 and 13). The composite bonded restorations were placed (Figures 14 and 15), and a final postoperative photo was taken (Figure 16).
Summary
Dental lasers are finding their way into everyday clinical use. Cosmetic dentists have known their advantages for years. They provide numerous advantages including the ability to shape, contour, and reestablish healthy gingiva with less bleeding; make final impressions the same time as treatment; decrease postoperative discomfort; and make healing predictable. Lasers will become the standard of care in cosmetic dentistry because of their advantages for the patient and clinician over steel blades and electrosurgery.20 With the addition of erbium lasers, procedures can be performed on the hard tooth structures that enhance bond strengths and virtually eliminate the need for anesthesia, because they are less invasive and traumatic than high-speed drills. All of these advantages will add up to patient and clinician satisfaction, but time must be spent researching the right laser for the practice and training the doctor and team. Only then will these advantages add up to delivering the best dentistry has to offer. ■
Acknowledgements ...
The author would like to thank Mr. Robert Hooyboer, Ceramique 2000 Dental Lab, Bristol, Tenn., for the excellent PFM restorations.
References
1 Rice JH. Laser use in fixed, removable, and implant dentistry. In: Convissar RA: Dent Clin North Am, Philadelphia, Pa., Saunders 2000; 44:767-778.
2 Rice JH. Erbium laser technology: today’s choice for tomorrow’s standard of care. WDJ July/Aug 2003; 1:10-21.
3 Maiman TH. Stimulated optical radiation in ruby. Nature 1960; 187:493-494.<
4 Goldman L, Gray JA, Goldman J, et al. Effects of laser impacts on teeth. J Am Dent Assoc 1965; 70:601-606.
5 Stern RH, Sognnaes RF. Laser beam on dental hard tissues. J Dent Res 1964; 43:873-876.
6 Manni JG: Dental applications of advanced lasers. Burlington, Mass., JGM Assoc 2000.
7 Myers TD, Myers WD, Stine RM. First soft-tissue study utilizing a pulsed Nd:YAG dental laser. Northwest Dent 1989; 68:14-17.
8 Sulewski JG. Historical survey of laser dentistry. In: Convissar RA, Dent Clin North Am, Philadelphia Pa., Saunders 2000; 44:717-729.
9 American National Standards Institute: American national standard for safe use of lasers in health-care facilities. Orlando, The Laser Institute of America 1996.
10 White JM, et al. Curriculum guidelines and standards for dental laser education. Proc SPIE, 1998; 3593:110-122.
11 Rice JH. Educate yourself in laser dentistry. Comp Esthet & Rest Practice Nov 2004.
12 Siminovsky GS. At your (regulatory) service. Wavelengths 2005; 13(3):10.
13 OSHA: Guidelines for laser safety and hazard assessment. U.S. Department of Labor: OSHA Instruction Publication 8-1.7. Directorate of Technical Support Aug 5, 1991.
14 Featherstone JD. Caries detection and prevention with laser energy. In: Convissar RA, Dent Clin North Am, Philadelphia, Pa., Saunders 2000; 44:955-966.
15 Gimbel CB. Hard tissue laser procedures. In: Convissar RA, Dent Clin North Am, Philadelphia, Pa., Saunders 2000; 44:931-948.
16 Moritz A, Schoop U, Gohorkhay K, et al. Procedures for enamel and dentin conditioning: a comparison of conventional and innovative methods. J Esthet Dent 1998; 10:84-93.
Janet Hatcher Rice, DDS
Dr. Rice, author, lecturer, and laser enthusiast, currently practices general dentistry in Bristol, Va./Tenn. She is the immediate past president of the Academy of Laser Dentistry and a past president of the American Association of Women Dentists. She lives on a 100-acre horse farm with her husband, Dr. John W. Rice. You may contact Dr. Rice at [email protected].
null
null
null
null
null
null
null
null
null