The growing popularity of mini-dental implants
By Raymond Choi, DDS, and Sarah E. Campbell
Dental implants, and their respective parts and tools, have become the fastest growing area in dentistry and the dental supply industry. Every year, more than 800,000 individual implants are placed in the United States and more than 1.8 million in the European Union. With an observed and projected annual growth rate of 15 to 20 percent, the dental implant market - in the United States alone - is forecasted to generate more than $900 million in revenues by 2009. Worldwide, the dental implant industry is expected to become a 1.2 to 1.4 billion-dollar market. While conventional, or regular diameter (3.0 mm diameter or greater), implants have historically dominated the bulk of the U.S. dental market, the demand for mini, or small diameter (3.0 mm or less), implants is growing substantially. This section of the industry is projected to grow by more than 35 percent in the coming years. The trend in implant dentistry is moving toward smaller and smaller devices, allowing more clinicians to place implants and proceed with noninvasive methods of serving the edentulous population. Many factors have contributed both to this trend and the seemingly exponential growth potential of the dental implant market.
Implant dentistry’s roots (no pun intended) stretch as far back as prehistoric man. There is early fossil evidence of attempts to recreate teeth using substances such as stone, ivory, and wood. As early as the 17th century, dentists were using tooth transplants to recreate the human mouth. Metal implants made their debut in the 20th century in many forms utilizing numerous types of metals including gold, lead, and CrCo.
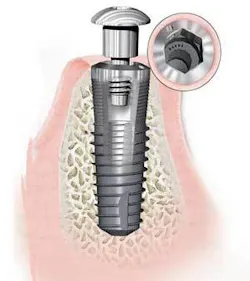
The metal implant has undergone many changes over time. In the 1940s, the metal implant was first born as a subperiosteal implant. This implant sat on top of the jawbone and directly below the periosteum. Next came the blades, followed closely by the staple form and the transmandibular form. Blade implants were flat, titanium implants with one or two metal prongs on one long side. A blade implant was placed in the jaw so that the prong(s) protruded into the mouth where they would support crowns or bridges. Transmandibular staple implants included two to four long metal screws, which were placed through the lower jawbone using a surgery starting from under the chin and extending into the mouth. A plate was fastened to the base of these screws and over the jaw but under the gum. These two to four screws served to anchor dentures, which may have been unstable without the implants. It was not until the 1960s that the root form (invented by Dr. Brånemark) made its debut. It mimicked the true form of a tooth and has shown the most success in osseointegration, a term coined by the Swedish orthopedist. Derived from the root form that was born in the 1970s, the mini-dental implant was being developed and modified as early as 1976. All of the implant forms mentioned above, except for the mini-implants, involved at least a two-stage, invasive surgical protocol. The typical surgical protocol for the mini-implants only requires a single, noninvasive procedure, and they can be immediately loaded following their implantation.
Throughout the history of dental implants, the trend toward smaller, yet fully functioning, implants has been apparent. This is partially responsible for the growth in popularity of mini-implants. Many other factors present themselves along with the dawn of mini-implants.
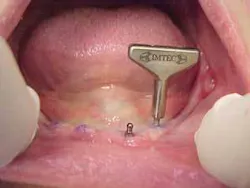
Mini-implants, which were first developed by Dr. Victor I. Sendax of New York in the early 1970s and later marketed by IMTEC Corporation in the 1990s, are just now beginning to realize their potential. Much of this is due to the sudden realization that these tools can have extreme benefits to patients. Due to their small size, mini-implants are logically less expensive than conventional implants. This lower initial cost is passed along to the patient through an overall more cost-effective procedure. This, though, may be the least among the list of benefits that has spurred the growth of mini-implants. With the Sendax IMTEC MDI (mini dental implant) surgical protocol, there are numerous physical benefits to the patient, which in the end are benefits to the clinician.
Clinicians working with MDIs have quickly learned that there is no waiting for the osseointegration process. Dentists have noted from positive patient feedback that MDIs cause much less discomfort than conventional implants, and patients are pleased with such a quick procedure. With thousands of MDIs in place, doctors have learned that the procedure is ideal for medically compromised, aging, and financially compromised patients. By introducing this tool into his Tustin, Calif., practice about seven years ago, co-author Dr. Choi has been able to assist numerous patients who were not good candidates for conventional implants or who could not afford conventional implant therapy.
During the evolution of the implant, and throughout the initial successful operations involving dental implants, many practitioners did not accept the modality as a form of dentistry. Since the dawn of the root-form implant, though, many different types of difficult oral treatments have been accomplished. This number has only increased since the birth of the mini-implant. While conventional implants (those ranging in diameters of 3 mm and up) require a substantial amount of good-quality bone for the implant to be successful, mini-implants can be placed in poorer quality bone and still have an extremely high success rate.
These minis, which were used initially for interim implants to support prostheses while the larger, conventional implants were in the “healing phase,” are now being seen as extremely useful in long-term applications. When minis were used as interim implants, the intent was to remove them when the larger implants were put into service. As might have been anticipated, when attempting to remove the interim mini-implants, practitioners found that they could not be removed because they had integrated into bone during the interim service period. Because of the fact that mini-implants showed such obvious evidence of osseointegration, as well as capability for immediate loading, some of the companies producing mini-implants have applied for approval of the small-diameter implants for long-term use.
IMTEC Corporation was the first company to receive FDA approval for long-term use of its MDI mini-dental implant devices in 2002, followed by BioHorizons and Dentatus USA. Nobel-Biocare was the latest entrant into the long-term-use, small-diameter implant market in 2004. With the approved use in long-term applications, the use of mini-implants is rapidly increasing and more research publications and clinical technique articles are becoming available. The introduction, approval, and continued observation of success of smaller-diameter mini-implants have stimulated use of implants in situations in which conventional implants could not have been used without grafting.
The popularity of single-stage surgeries will also help the mini-implant gain footing in the dental implant market. Nearly all indications for a mini-implant only require a single surgery or no actual surgery at all. Immediate functional loading accounted for only a small percent of the overall dental market (while it accounted for an overwhelming amount of the mini-implant market) in 2004. The lower cost of this technique is a result of shorter and more convenient treatment times, which will cause an increase in demand for items using this procedure over the next nine years. The mini-implant is not limited to use among oral surgeons or periodontists because of the ability to use it in a nonsurgical setting. As a rule, general practitioners are also able to perform the placement and restorative phases of implant dentistry. Since mini-implants have a simplified protocol, general practitioners are capable of placing them in their own clinics, giving them more freedom of control over their own patients and their course of treatment.
Long-term applications, along with their ease of use, have allowed the mini-implant market to grow like wildfire. The authors predict that in the future, dentists will see many more fixed applications beyond a long-term application for dentures and large prostheses. The authors point to how vital the mini-implant is becoming to cutting-edge orthodontia, as is the case with IMTEC’s ORTHO Implant.
The market for the mini-implant at present is filled with a small number of major competitors. IMTEC Corporation comprises more than 50 percent of the market share, while Dentatus USA, BioHorizons, and Nobel Biocare - along with a few other companies - make up the remaining sectors of the market. But changes are on the horizon. As more clinicians and educators begin to see the usefulness of the mini-implant, the market for the small-diameter implant will more than likely begin to reflect the market for conventional implants - with many different players taking part. The mini-implant, along with its numerous other advantages, will open the door for many patients to receive a quality-of-life improvement that was never possible with conventional implant therapy. There is no question that this is the next “big” little thing to happen in dentistry.
Raymond Choi, DDS, is a graduate of USC College of Dentistry and practices general dentistry in Tustin, Calif. Dr. Choi is a certified IMTEC MDI moderator, and conducts mini-residencies for dentists in his California clinic. He also conducts MDI seminars for IMTEC Corporation around the country and has lectured on the MDI and ENDURE implant lines in foreign locales for the company. Disclosure: Dr. Choi receives compensation from IMTEC for his services. He may be contacted via e-mail at [email protected] or via telephone at (714) 838-1111. Sarah E. Campbell is a marketing communications specialist with IMTEC Corporation in Ardmore, Okla. She may be contacted via e-mail at [email protected].