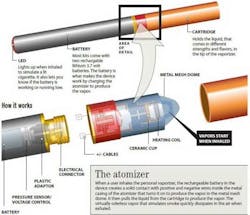
Health effects of e-cigarettes
E-cigarettes are battery-powered devices that convert nicotine into vapor.(1) The products are sold over the counter and are not subject to the same regulation as actual cigarettes. A 2011 survey showed that about 21 percent of smokers had used e-cigarettes at least once up from about 10 percent of smokers who took the same Web survey in 2010, according to a Centers for Disease Control and Prevention (CDC) study published online in February in Nicotine & Tobacco Research.(2,3)
About 7 percent of smokers who received the same survey in 2010 via postal mail also said they had tried e-cigarettes at least once. The Food and Drug Administration announced that the agency plans to propose regulating e-cigarettes as a tobacco product.(4)
Currently, e-cigarettes that are marketed for therapeutic purposes are regulated by the FDA Center for Drug Evaluation and Research (CDER). The FDA Center for Tobacco Products (CTP) currently regulates: cigarettes, cigarette tobacco, roll-your-own tobacco, and smokeless tobacco. FDA has stated its intent to issue a proposed rule that would extend FDA’s tobacco product authorities to products that meet the statutory definition of “tobacco product.”
For further details, please see the Unified Agenda entry describing this rulemaking.(8)
References
1. McGill N. Research on e-cigarettes examining health effects: Regulations due. http://thenationshealth.aphapublications.org/content/43/5/1.2.full.
2. King BA, Alam S, Promoff G, Arrazola R, and Dube SR. Awareness and Ever Use of Electronic Cigarettes Among U.S. Adults, 2010–2011. Nicotine Tob Res. First published online February 28, 2013. http://ntr.oxfordjournals.org/content/early/2013/02/20/ntr.ntt013.abstract?sid=d0df8d60-9de9-4c6d-89ab-07aed335f412.
3. Centers for Disease Control and Prevention. Quitting Smoking Among Adults—United States, 2001–2010. Morbidity and Mortality Weekly Report 2011; 60(44):1513–19 [accessed 2013 June 17]. http://www.cdc.gov/tobacco/data_statistics/mmwrs/byyear/2011/mm6044a2/intro.htm.
4. http://www.newsobserver.com/2013/05/19/2899478/fda-will-rule-none-too-soon-on.html.
5. http://www.fda.gov/tobaccoproducts/guidancecomplianceregulatoryinformation/ucm246129.htm.
6. http://www.lung.org/associations/states/florida/educational-programs/e-cigarettes.html.
7. http://voices.yahoo.com/american-lung-association-vs-electronic-cigarettes-5828424.html?cat=5.
8. http://www.reginfo.gov/public/do/eAgendaViewRule?pubId=201210&RIN=0910-AG38.