Regenerative endodontics
The management of the incompletely developed tooth with a necrotic pulp has always been a challenge within the field of endodontics. Cleaning and shaping these canals leaves the already thin dentin walls further susceptible to fracture. Additionally, the root canal system with an open apex is difficult to obturate, as there is a lack of a stop against which to condense material. Lastly, management of these cases is difficult as these cases often occur in children who have sustained trauma, where behavioral management and compliance may complicate the treatment.
Traditional means of treatment of necrotic, immature teeth included placement and changing of calcium hydroxide within the canal every three to six months for one year, possibly longer, to check for formation of an apical hard-tissue barrier. Recently, mineral trioxide aggregate (MTA) has been used to create an apical barrier to prevent extrusion of material when obturating these teeth to reduce treatment time to one or two visits. While both of these treatments have proven to be effective (1, 2), several aspects of these treatments are undesirable, including the need for several visits, patient compliance, and fairly recent evidence demonstrating that calcium hydroxide can cause decreased strength of dentin (3, 4), and — perhaps most importantly — there is limited potential for further development of the root. (5)
However, case reports published by Iwaya et al. (6) in 2001 and Banchs and Trope (7) in 2004 described a treatment modality that works to improve the limitations of the traditional means of treatment of the open apex. Based on these case reports, the area of regenerative endodontics was energized.
The premise for regenerative endodontics, quite simply, is to recreate the environment present when an immature tooth is avulsed. In the case of an avulsion, the pulpal tissues become necrotic not because of bacteria, but because they have been severed from their blood supply. If an immature tooth is replanted immediately due to the open apex, there is a chance that the vital tissues in the periapical area have the ability to grow into the canal space and regenerate, allowing for continued development of the root. In this scenario, the existing devitalized, but not infected, pulp tissue serves as scaffolding for the growth of new tissue. As long as the crown of the tooth is intact and there is no evidence of coronal leakage, the pulpal tissues can regenerate.
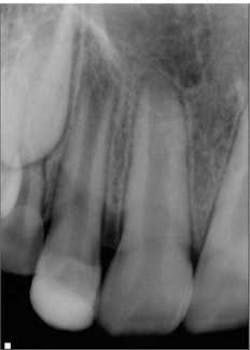
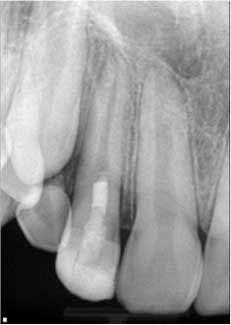
So how is the environment re-created? Let’s look at a case. Fig. 1 shows a lateral incisor that had sustained a concussive injury with an uncomplicated crown fracture. This 10-year-old boy presented to the office with complaint of pain, and swelling was present in the buccal vestibule. The tooth was diagnosed as having pulpal necrosis and an acute apical abscess. Due to the presence of an apical radiolucency and an immature apex, the regenerativeprotocol was chosen.Fig. 1: Tooth No. 7 showing incompletely formed apices and apical periodontitis. The patient also presented with pain to percussion and swelling.
At the first visit, the area was anesthetized, and the tooth was isolated with a dental dam and accessed. Upon access, necrotic tissue was noted in the canal. The canal was irrigated with 20 ml of 5.25% NaOCl, followed by 20 ml of saline, then 10 ml of 0.12% chlorhexidine. The canal system was dried, and a double antibiotic paste consisting of metronidazole and ciprofloxacin was placed in the canal system. Due to the swelling, the patient was given a prescription for amoxicillin and a analgesic medication. When the patient returned two weeks later, the symptoms and swelling had resolved.
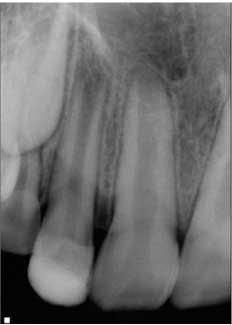
At the second visit, the tooth was anesthetized with an anesthetic that had no vasoconstrictors (to increase the likelihood of getting a blood clot in the tooth, Petrino et al., 2010), isolated, and accessed. The canals were irrigated with 20 ml of 5.25% NaOCl. After irrigation, the canal system was dried and a small, stainless steel hand file was used to lacerate the periradicular tissue to induce bleeding into the canal system. Once there was evidence of blood flow back into the canal, Collaplug (Zimmer Dental, Carlsbad, Calif.) was placed into the canal system to help create a barrier at the coronal portion of the canal. After formation of the blood clot, white MTA was condensed on top of the Collaplug to seal the canal system. The tooth was temporized, and the patient was instructed to return to his general dentist for restoration of the access (Fig. 2).Fig. 2: Tooth No. 7 after second visit showing placement of the MTA barrier.The patient was placed on recall and returned approximately eight months following treatment. The patient and parent reported that the tooth was not restored, but had been asymptomatic. Clinically, the tooth was asymptomatic, and radiographs showed signs of resolution of the apical radiolucency, increased thickness of the root walls, and apical closure. The patient was encouraged to have the access restoration restored. (Fig. 3) The patient returned at 15 months for his next recall examination, and the tooth had a permanent restoration. Clinically, the tooth was asymptomatic, and radiographs showed evidence of apical bone healing, increased thickness of the root walls, and apical closure. (Fig. 4)
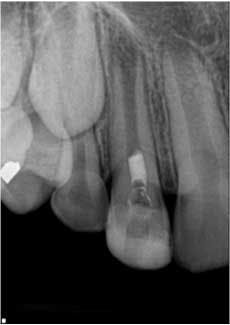
Fig. 3: Eight-month recall. Note the resolution of the apical periodontitis and continued development of the root walls and apical closure.
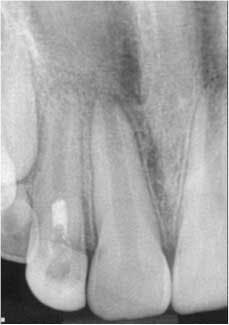
Fig. 4: 15-month recall. Note the continued apical development and intact and uniform PDL and LD. Also note the hard-tissue barrier apical to the MTA.
This case, as well as other case reports and case series, demonstrates the tremendous potential of regenerative endodontics (5, 8, 9). Since the description of this technique in 2001 and 2004, there have been considerable advancements in the area of regenerative endodontics, including a standing committee on Regenerative Endodontics within the American Association of Endodontics (AAE). Examples of regenerative endodontic research include identification of stem cells in the apical papilla subjacent to immature teeth, development of scaffolds on which to regenerate tissue, and additional or more beneficial means of cleaning the canal system. Additionally, a database of cases has been established by the AAE to aid in developing research to improve the recommendations for regenerative endodontic treatment. While this type of treatment is beneficial and has demonstrated successful outcomes, unfavorable outcomes can occur. Some outcomes include coronal discoloration, poor or no root development, or difficulty in inducing bleeding (10, 11). Due to the research, case reports, and collaboration through the database, treatment considerations are regularly being updated. The up-to-date treatment considerations can be found here. (12).
In conclusion, regenerative procedures are a departure from traditional endodontic techniques in that they only use chemical means of cleaning the canal system(s), induction or placement of scaffolds, vs. the traditional means of chemomechanical disinfection of the canal system. Regenerative endodontic treatments hold promise that a functional pulp-dentin complex can be regenerated for our patients. While a majority of the regenerative endodontic procedures are being performed on immature teeth, investigators are now researching whether these types of treatment can be done in fully developed teeth. It is an exciting time in the field of endodontics. In a little more than 10 years, regenerative endodontics has made a significant impact. Perhaps within another 10 years, these types of procedures can be applied to developed teeth and practitioners can help patients regenerate a functional pulp dentin complex for our patients.
Acknowledgements:Dr. Petrino would like to thank Drs. Alan S. Law, John Bogle, and Scott McClanahan for their help in preparation and reviewing of this manuscript.
References
1. Ghose LJ, Baghdady VS, Hikmat BYM. Apexification of immature apicies of pulpless permanent anterior teeth with calcium hydroxide. J Endod 1987;13:285-290.
2. Witherspoon DE, Small JC, Regan JD, Nunn M. Retrospective analysis of open apex teeth obturated with mineral trioxide aggregate. J Endod 2008;34:1171-1176.
3. Andreasen JO, Farik B, Munksgaard EC. Long-term calcium hydroxide as a root canal dressing may increase risk of root fracture. Dent Traumatol 2002;18:134-137.
4. Batur YB, Erdemir U, Sancakli S. The long-term effect of calcium hydroxide application on dentin fracture strength of endodontically treated teeth. Dent Traumatol 2013;29:461-464.
5. Nagy MM, Tawfik HE, Hashem AAR, Abu-Seida AM. Regenerative potential of immature permanent teeth with necrotic pulps after different regenerative protocols. J Endod 2014;40:192-198.
6. Iwaya S, Ikawa M, Kubota M. Revascularization of an immature permanent tooth with apical periodontitis and sinus tract. Dent Traumatol 2001;17:185-187.
7. Banchs F, Trope M. Revascularization of immature permanent teeth with apical periodontitis: new treatment protocol? J Endod 2004;196-2004.
8. Kahler B, Mistry S, Moule A, Ringsmuth A, Case P, Thomson A, Holcombe T.DOI: http://dx.doi.org/10.1016/j.joen.2013.10.032 Revascularization outcomes: A prospective analysis of 16 consecutive cases. J Endod 2013;40:333-
9. Bose R, Nummikoski P, Hargreaves K. A retrospective evaluation of radiographic outcomes in immature teeth with necrotic root canal systems treated with regenerative endodontic procedures. J Endod 2009;35:1343-1349.
10. Nosrat A, Homayounfar N, Oloomi K. Drawbacks and unfavorable outcomes of regenerative endodontic treatments of necrotic immature teeth: A literature review and report of a case. J Endod 2012;38:1428-1434.
11. Petrino JA, Boda KK, Shambarger S, Bowles WR, McClanahan SB. Challenges in regenerative endodontics: A case series. J Endod 2010;36:536-541.
12. Law AS. Considerations for regeneration procedures. J Endond. 2013;DOI: http://dx.doi.org/10.1016/j.joen.2012.11.01939:S44-S56.