FDA clears MicroDental’s MicrO2, the first CAD/CAM-milled sleep apnea device
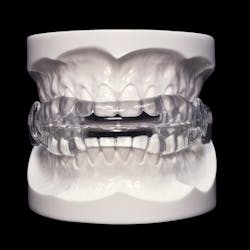
MicroDental Laboratories announced that the Food and Drug Administration (FDA) has cleared the company’s MicrO2 obstructive sleep apnea device. The first CAD/CAM-milled obstructive sleep apnea device, the MicrO2 offers dentists a new and innovative option for treating the growing number of patients diagnosed with obstructive sleep apnea (OSA).
“Dentists are the front line for screening and managing patients with obstructive sleep apnea,” noted Kim Bradshaw, CEO and President of MicroDental Laboratories. “Many of our partner dentists asked for a small, precise, and easy-to-use obstructive sleep apnea device. The MicrO2 device has been designed, from the very beginning, with these goals in mind.”
ADDITIONAL READING | Marketing for patients with obstructive sleep apnea (OSA): catchment segmentation with coordinated touch points
“I believe the MicrO2 will revolutionize dental sleep medicine,” stated Dr. Bill Dickerson, DDS, FAACD, LVIM. “I can honestly say that I don’t think there is a better appliance out there. With its [lingual-free] design, airway constriction preventing fins, and unobtrusive size, this device is a winner.”
Featuring a patent-pending design and a precision-milling production process, the MicrO2 is able to utilize a PMMA material that is cured under controlled conditions. These aspects allow the MicrO2 to be small yet strong, precise, and predictable. The MicrO2 also features a unique approach to titration that is intended to make the device easier to use for the dentist and the patient.
“The MicrO2 does not require patients to manage any mechanical adjustments, such as turning screws or changing positions with elastics. Instead, the MicrO2 comes with one pair of upper and lower arches set to the prescription and a second pair set in any amount of advancement increments. Advancements are easily achieved by simply removing the current upper or lower arch device and inserting the next advancement device in the MicrO2 series,” commented Laura Sheppard, Senior Director of Compliance and Regulatory Affairs for MicroDental.
Obstructive sleep apnea is one of the fastest developing frontiers in dentistry. A report by the Institute of Medicine estimates that 50 to 70 million Americans suffer from chronic sleep disorders. More than 18 million Americans have obstructive sleep apnea, which has been linked with snoring, memory loss, headaches, depression, and an increased risk of stroke, heart problems, and motor vehicle accidents.
ADDITIONAL READING | Hidden dental dangers of undiagnosed obstructive sleep apnea
“MicroDental Laboratories has a legacy of partnering with dentists who are committed to enhancing the lives of their patients,” noted Len Liptak, Vice President of Sales and Marketing for MicroDental. “With our FDA-registered MicrO2 device, we look forward to partnering with these dentists as they manage the growing number of patients who are diagnosed with this serious condition.”
***
Editor's Note: Have you used this product? Tell us what you think! Comment below, or join the discussion on DentistryIQ's Facebook and Twitter pages. If you are a manufacturer or distributor and are interested in having DentistryIQ review this product, including a Dental Economics "Pearls for Your Practice" feature by Dr. Joshua Austin, find our contact information here.